Oxygen Electron Configuration (O) with Orbital Diagram
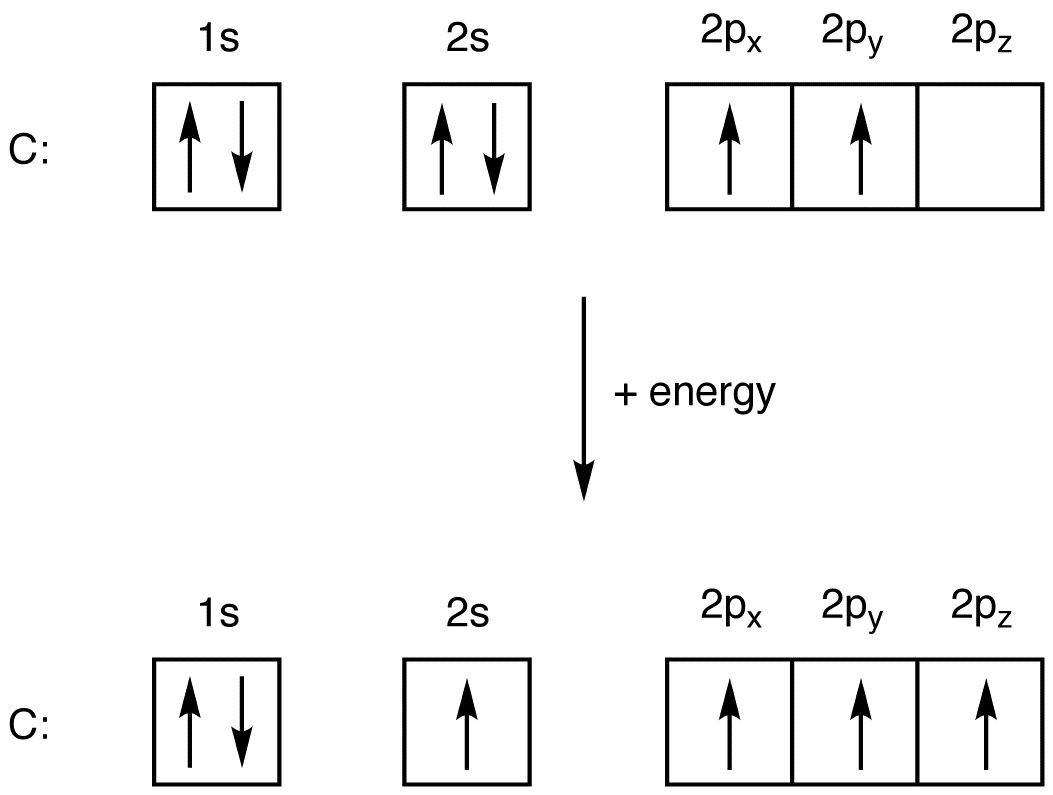
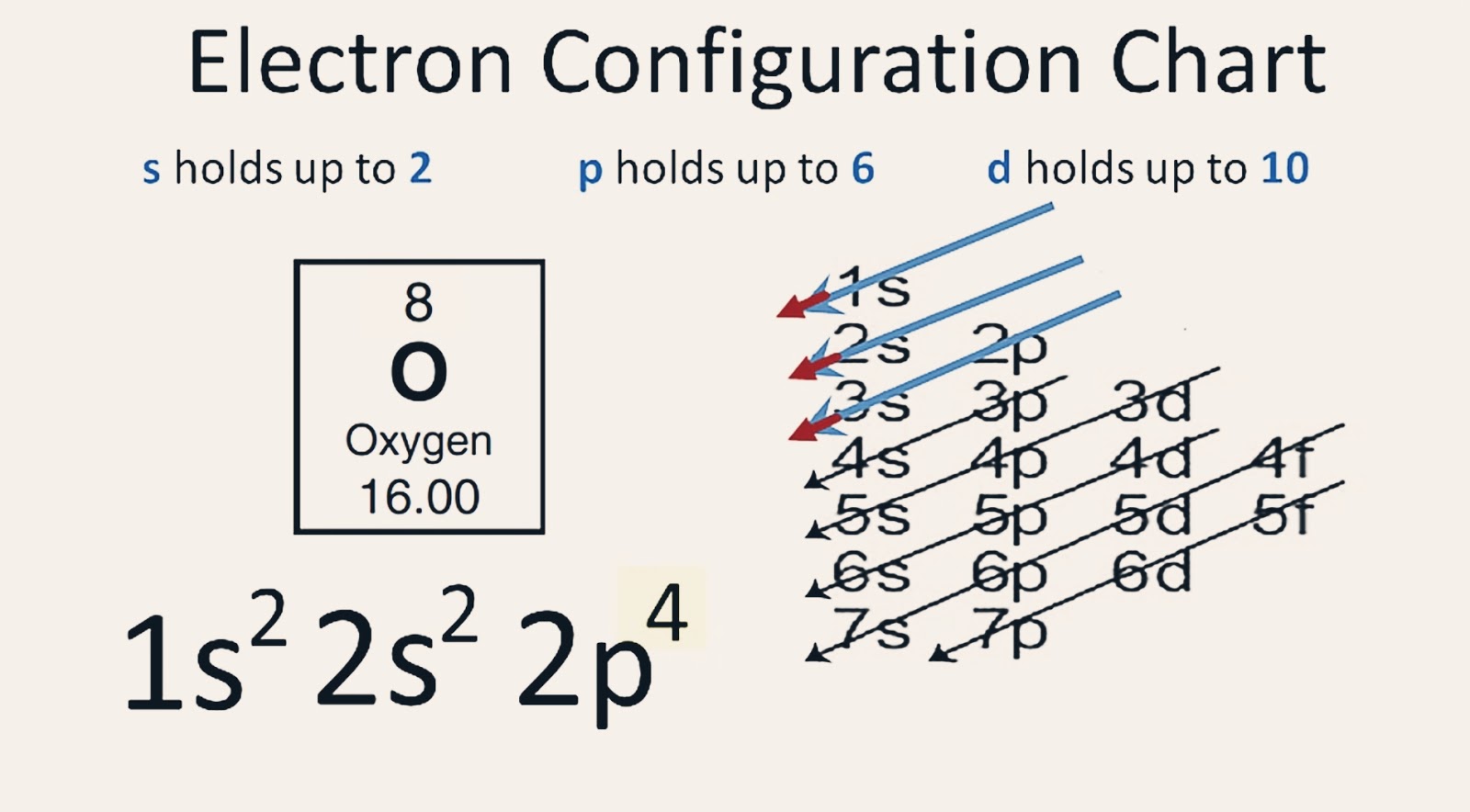
Based on the order of fill above, these 8 electrons would fill in the following order 1s, 2s and then 2p. So Oxygen's electron configuration would be O 1s 2 2s 2 2p 4. Special Cases. Configurations of ions present a special case of electron configuration and also demonstrate the reason for the formation of those ions in the first place.

Oxygen Atom Science Notes and Projects
Oxygen Electron Configuration: O is an odourless, colorless, reactive gas. Its atomic number is 8 and it the life-supporting component of the air. It forms almost 20 percent of the earth's atmosphere. It is also the most abundant element in the earth's crust which is found mainly in the form of silicates, oxides, and carbonates.
Diagram representation element oxygen Royalty Free Vector
Electron Configuration Chart of All Elements (Full Chart) March 23, 2023 by Jay Electron configuration chart of all Elements is mentioned in the table below. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Free Gift for you: Interactive Periodic Table

Electronic configuration of the oxygen atom Download Scientific Diagram
Electronic configuration: The arrangement of electrons into the orbitals of an atom using some fundamental principle is called its electronic configuration. Electronic configuration of Oxygen: The electronic configuration of Oxygen is 1 s 2 2 s 2 2 p 4. Oxygen requires two electrons to attain noble gas configuration. Suggest Corrections 24

Isotopes oxygen structure atome labeled Royalty Free Vector
We can write the configuration of oxygen's valence electrons as 2s²2p⁴. Created by Sal Khan. Questions Tips & Thanks Want to join the conversation? Sort by: Top Voted 419522 6 months ago At 4:26 , Sal says "In most cases, your valence electrons are going to be your outermost electrons."
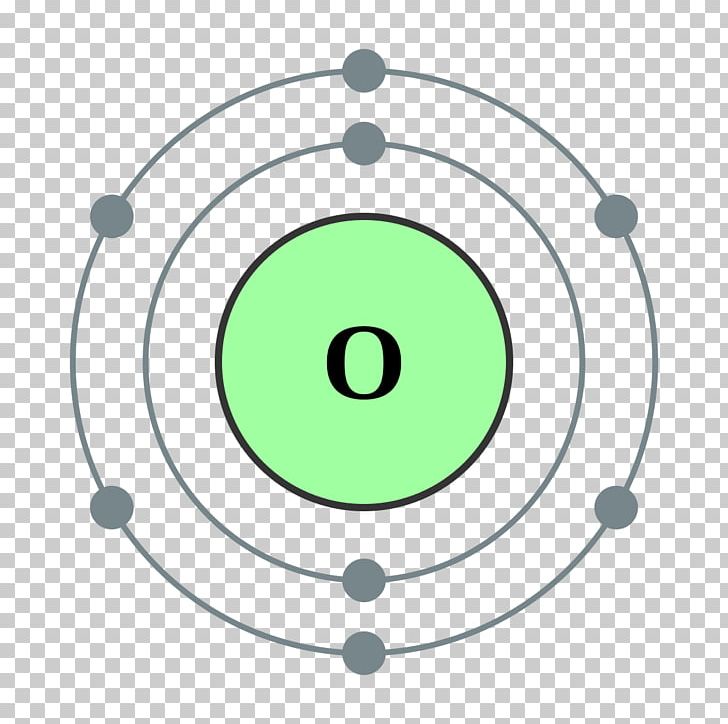
Atomic Number Oxygen Bohr Model Chemical Element, PNG, 1024x1024px
Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons.

Oxygen Bohr Model (Diagram, Steps To Draw) Techiescientist
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,
Oxygen(O) electron configuration and orbital diagram (2022)
Example 1.6. 3: Carbon and Oxygen. Consider the electron configuration for carbon atoms: 1s 2 2s 2 2p 2: The two 2s electrons will occupy the same orbital, whereas the two 2p electrons will be in different orbital (and aligned the same direction) in accordance with Hund's rule. Consider also the electron configuration of oxygen.
Bohr Model Chemical Element Oxygen Atomic Theory PNG, Clipart, Angle
In order to write the O electron configuration we first need to know t.

Symbol and electron diagram for Oxygen Royalty Free Vector
The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium. The remaining electron must occupy the orbital of next lowest energy, the 2 s orbital (Figure 8.3. 3 or 8.3. 4 ). Thus, the electron configuration and orbital diagram of lithium are:
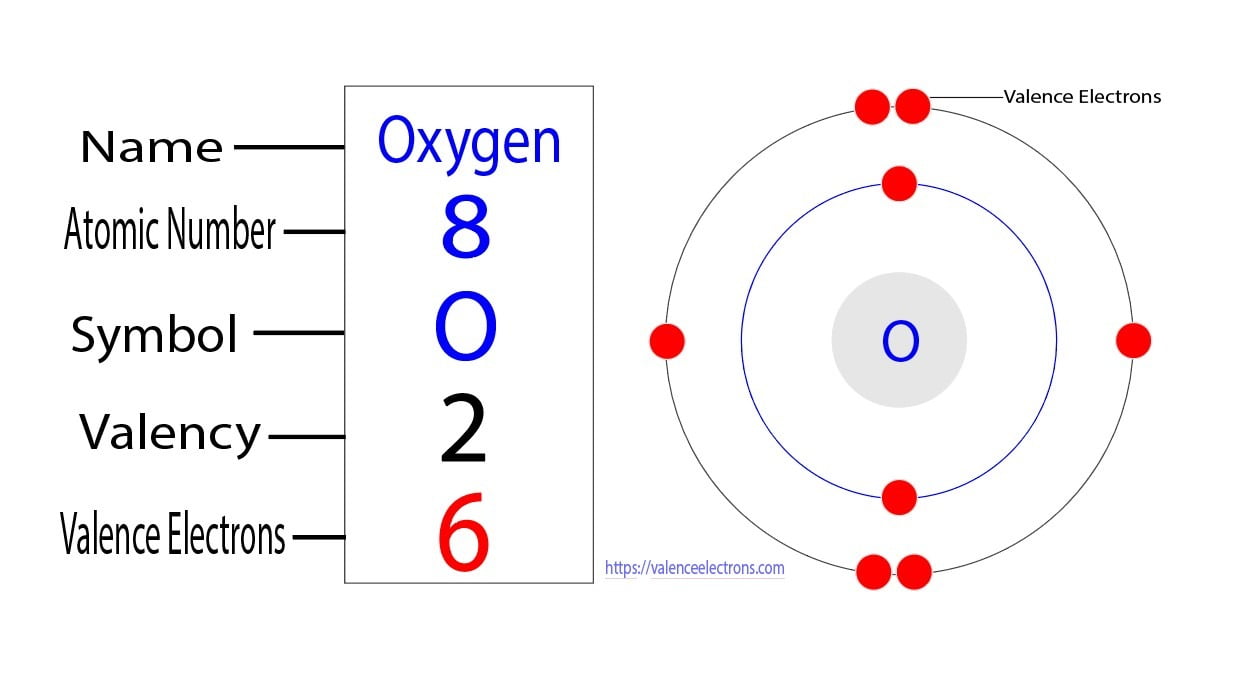
How to Find the Valence Electrons for Oxygen (O)?
The arrangement of electrons in oxygen in specific rules in different orbits and orbitals is called the electron configuration of oxygen. The electron configuration of oxygen is [ He] 2s 2 2p 4, if the electron arrangement is through orbitals. Electron configuration can be done in two ways. Electron configuration through orbit (Bohr principle)

Electron configuration of oxygen ion Lousiana
O 2- Electron Configuration (Oxide Ion) Wayne Breslyn 728K subscribers Join Subscribe Subscribed 799 130K views 4 years ago In this video we will write the electron configuration for O 2-, the.
What Is the Oxygen Electron Configuration(O)?
Let's find the electron configuration of Oxygen! A single oxygen atom has 8 protons and 8 electrons, but how do we know where Oxygen puts its electrons, in w.

The electron configuration of oxygen is 1s2,2s2 2p4. Science
Members of a group typically have similar properties and electron configurations in their outer shell. Period A horizontal row in the periodic table. The atomic number of each element increases by one, reading from left to right.. Oxygen accounts for about 23% of the atmosphere's mass with pairs of oxygen atoms stuck together to make.

oxygen atom Chuba Oyolu's Portfolio
If we look at the element after nitrogen in the same period, oxygen (Z = 8) its electron configuration is: 1s 2 2s 2 2p 4 (for an atom). Oxygen has one more electron than nitrogen and as the orbitals are all half filled the electron must pair up. Occupation of Orbitals.
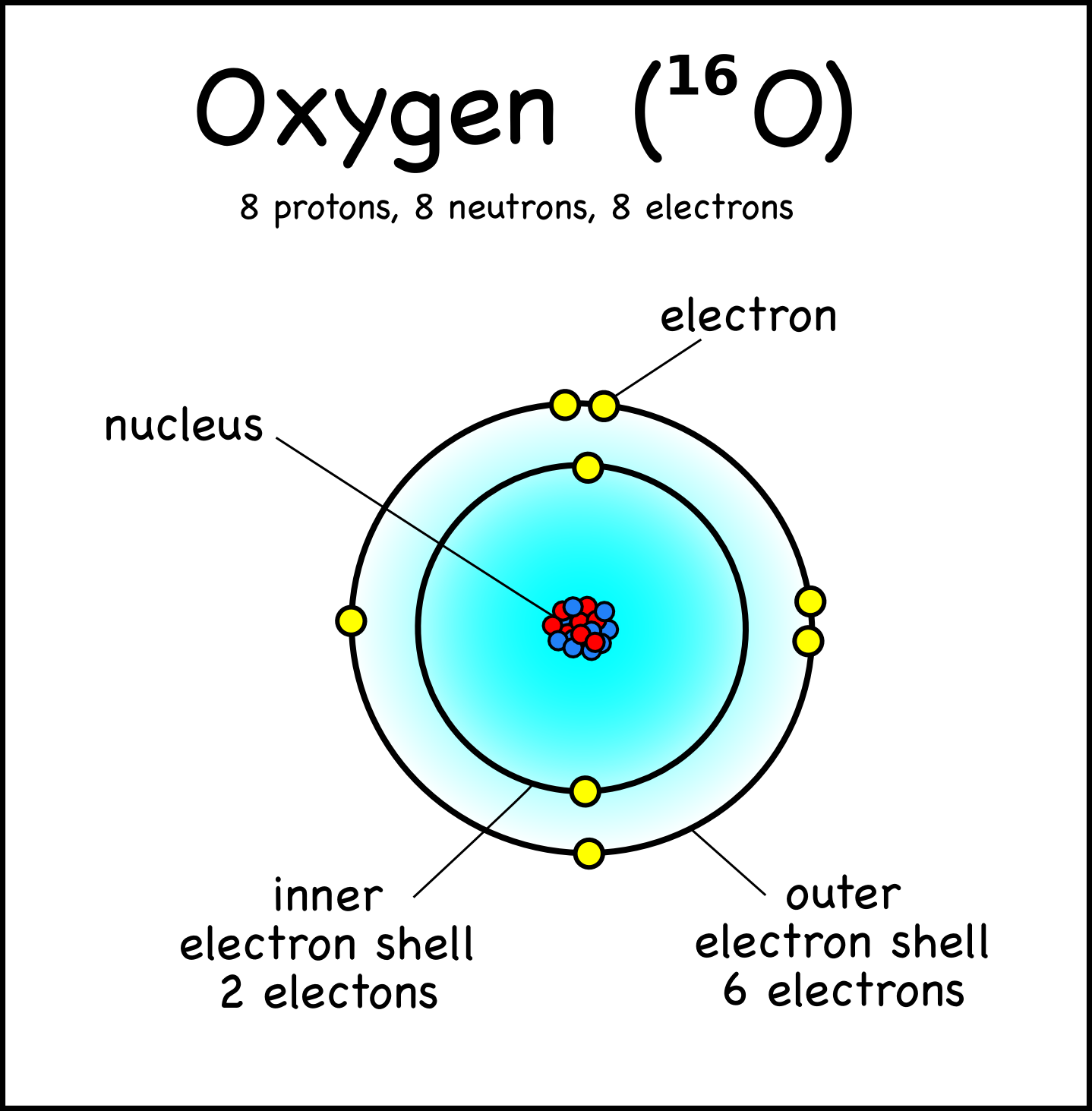
atoms and molecules Page 3 Montessori Muddle
In this case, 2+2+6+2+6+2+10+6+2+1= 39 and Z=39, so the answer is correct. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z = 83). The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3.